For those of you working to the orange guide and/or the guidance in the European Pharmacopoeia, you will notice the absence of specific guidance on how to monitor your cleanrooms. The USP <797> however, does give specific advice on what media to use and specifies that surface sampling must be supplemented with additives to neutralise the effects of disinfectants. To be more precise, the residue left by disinfectants post cleaning affects the efficacy of the growth medium, possibly masking the presence of viable micro-organisms, especially those on surfaces.
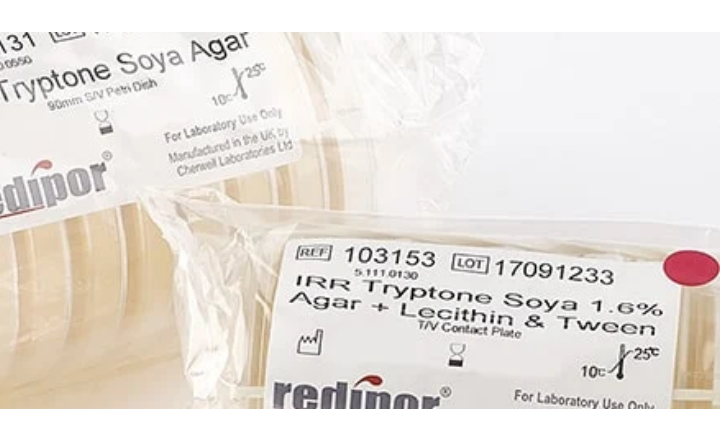
At Cherwell Laboratories our Redipor range of irradiated media can be purchased with several different combinations of neutralisers. The question is which neutralisers shall I use? Although the European Pharmacopoeia has little information on environmental monitoring it does specify in another section (2.6.12 Microbial examination of non-sterile products: Microbial enumeration tests) which neutralisers to use with specific interfering substances. Taken from the EP table 2.6.12 – Common neutralising agents for interfering substances:
From the above list, you will more than likely recognise that Lecithin and Tween 80 will neutralise the most common biocides in cleanroom disinfectants. The increasing use of chlorine-based sporicidal disinfectants is driving more people to choose the 4 neutraliser versions for surface samples.
70% alcohol is used often during aseptic operations but does not require specific neutralisers. It has no dry residue and if vapour or microdroplets contact the agar surface, the dilution would eliminate the biocidal activity.
Hydrogen peroxide vapour is increasingly popular as a sporicidal agent for decontaminating isolators and material transfers. GMP requires that it is removed completely by aeration prior to aseptic operations. It also has no dry form, so no residue, and dissociates rapidly to water and oxygen on contact with organic material including agar. However, it is important that agar is not exposed during the gassing phase of a H2O2 decontamination cycle when the agar might absorb sufficient H2O2 to produce toxic radicals or oxidise nutrients. The plates should have been irradiated in a validated impermeable wrap to protect them in that process, as used in the Redipor Barrier Pack.
If you have any technical questions regarding the use of neutralisers in growth media or have any other questions regarding Cherwell Redipor irradiated media please contact us using the request information button below.