RNAssist (Cambridge UK) and Rapid Labs Ltd (Colchester, UK) have launched the virusPHIX™ range of virus inactivating and RNA stabilising transport media, following successful SARS-CoV-2 inactivation testing carried out by Public Health England (PHE).
virusPHIX™ is a novel RNA stabiliser and virus inactivation medium based on technology developed by RNAssist Limited (Cambridge, UK). It has been shown to stabilise viral RNA for up to 1 year at 20°C and has successfully inactivated all tested viruses and bacteria to date, including SARS-CoV-2, Hepatitis B, Vaccinia, FIV, BVDV, Dengue, Zika, Influenza A and M. tuberculosis.
Unlike other virus transport mediums, virusPHIX™ does not contain guanidine, a highly toxic substance that can produce hydrogen cyanide when mixed with household cleaning products such as bleach.
virusPHIX™ is a completely non-toxic, non-volatile reagent that can be used safely in a home testing environment and shipped for PCR analysis. The virusPHIX™ range of products are manufactured and sold worldwide by Rapid Labs Limited (Colchester, UK).
With over 200 evaluators worldwide, virusPHIX™ is a popular choice for RNA stabilisation thanks to its compatibility with most front-end RNA purification kits.
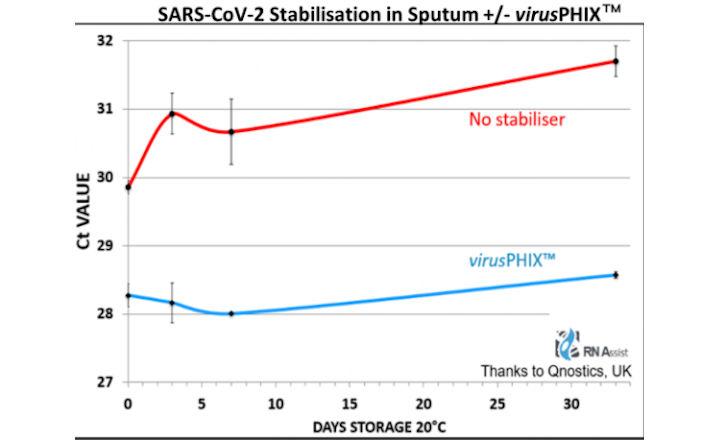
Public Health England and The Roslin Institute have demonstrated inactivation of SARS-CoV-2 within 10 minutes at 20°C. Qnostics UK has also demonstrated SARS-CoV-2 RNA stabilisation for 33 days at room temperature, and 12-month stability of 2 HCV strains at 20°C.
The virusPHIX™ range features 3 reagents suited to different applications:
- virusPHIX+™ for swab and saliva testing
- virusPHIX-LV™ (lower viscosity) for swab testing on automated liquid handling platforms (Ortho, Hamilton and Tecan)
- virusPHIX-P9™ for swab and saliva testing, especially when it is necessary to add sample directly into an RT-LAMP, LamPORE or RT-PCR assay without RNA purification.*
*virusPHIX-P9™ has been demonstrated to be directly compatible with LAMP assays.
Please use the 'Request Information' button provided below to find out more.
Note: This content has been edited by a rapidmicrobiology staff writer for style and content.