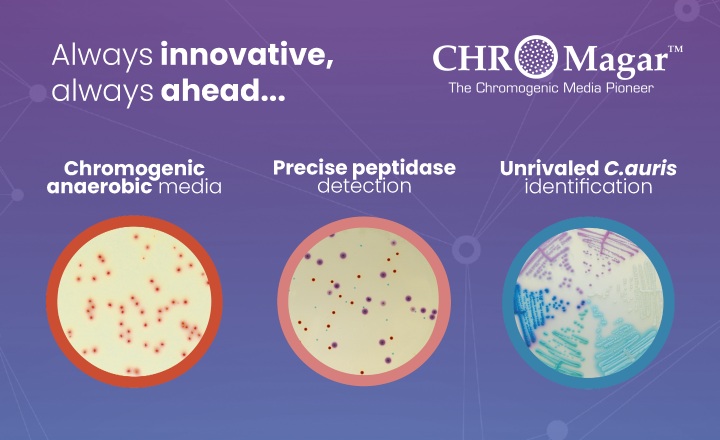
ATCC® has taken the guesswork out of choosing control strains for Clostridium difficile research by creating a uniquely-designed panel of clinical isolates, including:
- Representatives of each known toxinotype The ATCC Clostridium difficile Panel (ATCC® MP-4™) is comprised of 8 clinical isolates representing each one of the known C. difficile toxinotypes including types 0, IIIb, IIIc, (tcdA-, tcdB-), V, VIII, XII, and XXII.
- Improved characterization profiles Each strain in the ATCC® MP-4™ Panel is tested for toxinotype, the presence or absence of binary toxin, Enzyme Immuno Assay (EIA) results, and ribotype. A complete listing of all Clostridium difficile strains tested by ATCC is also available.
- Strains indicated in major outbreaks Significant strains incorporated into the ATCC Clostridium difficile Panel (ATCC® MP-4™) include an epidemic strain implicated in numerous outbreaks across North America ( ATCC® BAA-1870™) and a toxinotype V strain ( ATCC® BAA-1875™) strongly associated with food-producing-animal infections in Europe and the United States.
ATCC also offers Custom DNA Extraction for each microbial strain included in the panel, as well as normal human foreskin fibroblasts that can be used in Clostridium difficile cytotoxicity neutralization assays, including:
- ATCC® CRL-2429™ (CCD-1112Sk)
- ATCC® CRL-2522™ (BJ)
- ATCC® SCRC-1041™ (HFF-1)
- ATCC® PCS-201-010™ (Primary neonatal foreskin fibroblasts)
ATCC has been the globally trusted resource for biological materials and reference reagents for over 85 years. ATCC cultures provide consistency in research applications, ensuring reproducible and reliable data.
Please note : Any products described on this page are
for Research Use Only and not intended for clinical diagnostic procedures unless otherwise stated.