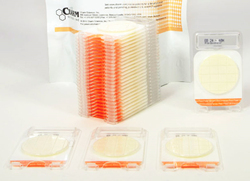
 Charm Sciences have introduced a new AOAC – OMA-approved Peel Plate® EB Microbial Test for use in the poultry processing industry.
Charm Sciences have introduced a new AOAC – OMA-approved Peel Plate® EB Microbial Test for use in the poultry processing industry.
The new test allows processing facilities to perform indicator tests onsite and provides quick, clear, and easy-to-interpret results.
The Peel Plate EB Microbial Test is validated to be used with either neutralizing buffered peptone water (nBPW) or buffered peptone water. Federal regulators now exclusively test with nBPW, making Peel Plate EB Microbial Test the ideal testing solution for poultry processors who want to align their bacterial sampling processes with rapid and clear test results for implementing any remediation processes.
“We are pleased to be introducing this new Peel Plate EB Microbial Test to market, as it will significantly assist the poultry processing industry’s efforts to ensure the safety of poultry products entering our food system,” said Lindsey Ross -Technical Sales Representative. “Poultry plants that use our new Peel Plate EB Microbial Test are aligning their indicator methodology with the system and standards used by federal regulators, taking the guesswork out of comparing different sampling and testing methods. It is a quicker, simpler test to perform, it can be done onsite, and results remove the ambiguity of test interpretation—meaning companies can address problems soon after they are identified.”
By using a test that aligns with regulatory testing standards, facilities are removing uncertainty when determining when and if remediation steps are necessary—without waiting on the long turnaround time of pathogen testing. The Peel Plate EB Microbial Test can be performed onsite, and provides clear, easily understood results fast.
Visit www.charm.com/products/test-and-kits/microbial-detection-tests/peel-plate-eb-microbial-test