The Monocyte Activation Test (MAT) is currently the only in vitro method designed for the detection of both endotoxins and non-endotoxin pyrogens (NEPs). Using the Mono-Mac-6 (MM6) cell line as a source of monocytes, the PyroMAT® test mimics the human immune reaction to pyrogens to measure the pyrogenic effect of parenteral products.
This new solution has been evaluated with samples such as albumin, fetal bovine serum, and vaccines, and our latest application note demonstrates the effectiveness of the PyroMAT® system with hormone-based drugs.
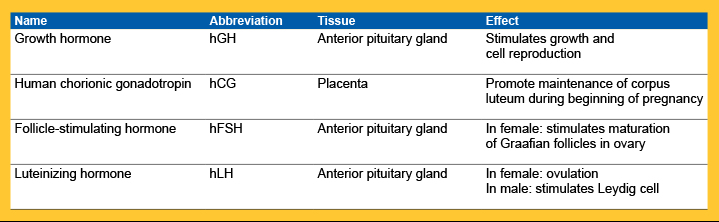
In this study we describe a range of suitability tests performed on the following selection of injectable hormone drugs.
To read the method, results and analysis, download our application note.
With the PyroMAT® system, we offer a robust and sensitive solution for pyrogen detection supported by international regulations and guidelines to reduce animal consumption for pyrogen tests.