Aflatoxins are toxic metabolites produced by a variety of molds such as Aspergillus flavus and Aspergillus parasiticus. They are carcinogenic and can be present in grains, nuts, cottonseed and other commodities. Crops may be contaminated by one or more of the four sub-types of aflatoxin: B1, B2, G1 and G2. Aflatoxin B1 is the most toxic and frequently detected form and implicated in human health disorders primarily hepatocellular carcinoma and aflatoxicosis. When cows consume Aspergillus spp. contaminated feeds, aflatoxin B1 is converted to aflatoxin M1, which is subsequently secreted in the milk of lactating cows. Aflatoxin M1 is quite stable towards normal milk processing methods such as pasteurization, and it may persist into final products for human consumption. Most government agencies worldwide have regulations regarding the amount of aflatoxins allowed in human and animal foodstuffs. In the US, the allowable limit is > 0.5µg/L or 500 parts per trillion (ppt) and in the European Union (EU), this limit has been set at > 0.05µg/L or 50 ppt. Hygiena’s Helica Biosystems Division has been supplying dairy markets worldwide >15 years with a high sensitivity format with a detection range of 5 – 100 ppt and assay time of 2.5 hours. An independent publication in 2019 showed this assay to be one of the best for recovery, precision and accuracy.
Continuous development has resulted in high sensitivity Aflatoxin M1 with an extended detection range of 5 – 500 ppt and significantly reduced assay time of 70 minutes. This new format adds versatility to the product making it useful as a single universal ELISA kit that can meet all regulatory requirements. It also allows detection of high levels of aflatoxin M1 up to 500 ppt without any additional dilutions.
The assay is a competitive immunoassay (ELISA). The procedure includes pipetting of sample or standard in the antibody coated wells, washing the wells, pipetting a mixture of HRP conjugate and sample or standard, washing again, adding substrate solution and stop solution. The wells are read on a microplate reader set at a wavelength of 450 nm. The optical densities (OD) of the samples are compared to the OD of the kit standards and an interpolated result is determined.
An additional independent third party laboratory was conducted in 2020 to verify the performance of Aflatoxin M1 ULTRA kit and compare it with another commercially available kit. The study validated the broad dynamic range 5 - 500 ppt for milk samples (without a sample dilution) with detection at 5 ppt in milk. Recovery (typically 96 – 118%), precision and accuracy ( -1.5 to 12.6) was better than competitor kit and was also better than that of the 2019 publication. Our research shows that Hygiena Aflatoxin M1 ULTRA ELISA kit has the widest and most sensitive quantitation range available in the market.
In contrast to other kits available in the market, Hygiena Aflatoxin M1 ULTRA has extremely low cross reactivities (less than 0.1%) to Aflatoxin subtypes (M2, B1, B2, G1 and G2) that could cause false positive results due to coexistence of those subtypes. This is an important benefit in testing dairy products (milk and yoghurt) containing added grains or nuts (barley, oat, wheat or walnut) that could be potentially be contaminated with aflatoxin B1, B2, G1 and G2.
The new Aflatoxin M1 ULTRA ELISA complies with ISO 14675* guidelines and significantly exceeds these guidelines in the following ways and is posed to be the Aflatoxin M1 ELISA method of the future:
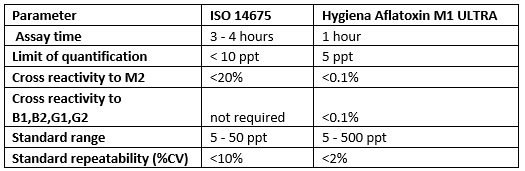
* ISO 14675:2003 [IDF 186:2003] Milk and milk products — Guidelines for a standardized description of competitive enzyme immunoassays — Determination of aflatoxin M1 content. By: Technical Committee : ISO/TC 34/SC 5 Milk and milk products
The Hygiena Aflatoxin M1 ULTRA is now available for distribution worldwide together with a range of other mycotoxin ELISA kits validated for several food types such as coffee, spices, wine and other.
Please contact Helica-ts@hygiena.com for additional information.