For the antimicrobial susceptibility testing of fosfomycin IV, EUCAST recommends;1
| Indication | MIC breakpoints (mg/L) | Disk content (μg) | Zone diameter breakpoints (mm) | ||
| S ≤ | R > | S ≥ | R < | ||
| Fosfomycin IV | 32 | 32 | 200 | 24 | 24 |
The European Committee on Antimicrobial susceptibility Testing (EUCAST) has released its recommendations for Fomicyt®IV (fosfomycin IV) testing with Enterobacteriaceae. These new recommendations, for both MIC determination and disk diffusion methods, provide clarity and critical guidance when testing fosfomycin IV for use against hospital-acquired pathogens in the Enterobacteriaceae species, including Escherichia coli.1
Intravenous fosfomycin IV is an effective antibiotic treatment indicated for infections when commonly recommended first-line antibiotics are considered inappropriate. Intravenous fosfomycin has been available for some time, but is not commonly used in UK hospitals due in part to lack of specific EUCAST guidance on testing.
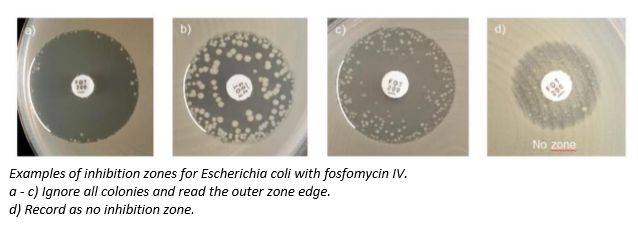
“Validated using E.coli as per standard practice, EUCAST’s recommendation provides microbiologists with zone diameter breakpoints to apply to E.coli”, explained Dr Mandy Wootton, Secretary, Standing Committee on Susceptibility Testing, British Society for Antimicrobial Chemotherapy (BSAC). “What is crucial is the clarity provided by the EUCAST guidance around ignoring the colonies inside the zone of inhibition. This is a very positive and a welcomed update as it allows microbiologists to confidently recommend fosfomycin IV, widening the treatment options available to fight hospital-acquired infections”.
EUCAST’s update provides clinicians with confidence through testing of fosfomycin IV as an all-important additional option when treating hospital-acquired bacterial infections, allowing doctors to keep last-line carbapenems in reserve for the most severe of infections and align to QIPP and CQUIN targets, and the UK 5 year Antimicrobial Resistance strategy.
Fosfomycin is a phosphonic acid derivative and is unique in its composition, being chemically unrelated to any other known antibiotic. With a mechanism of action unlike all other antibiotic classes, fosfomycin IV reduces the risk of cross-resistance and has synergistic or additive effects in combination with many other antibiotics. Fosfomycin IV targets a wide range of Gram-positive and negative bacteria and with a molecular mass of 138, is one of the smallest antibiotics in clinical use, enabling improved penetration well into interstitial fluid and tissues, achieving bactericidal levels. Fosfomycin IV has proven efficacy in critically ill patients with multi-drug resistant infections as part of combination therapy, with low in-hospital antimicrobial resistance demonstrated by high exposure rates over time.
Reference:
1. EUCAST, the European Committee on Antimicrobial Susceptibility Testing. Breakpoint tables for interpretation of MICs and zone diameters. Version 7.0, 2017. www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/Breakpoint_tables/v_7.0_Breakpoint_Tables.pdf



















