E.coli O157 in Raw M...
ABRx™ Antibiotic R...
11th October 2016 Content supplied by: BD Diagnostic Systems
New BD Veritor™ Plus System for Secure, Traceable POC
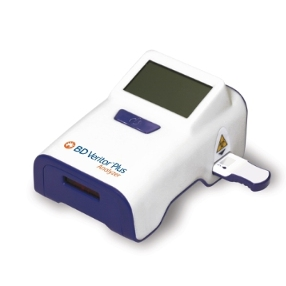 BD (Becton, Dickinson and Company) have launched its next generation wireless rapid diagnostic system for detection of influenza A and B, respiratory syncytial virus (RSV) and group A strep, with new traceability and secure patient health record documentation features and functionality.
BD (Becton, Dickinson and Company) have launched its next generation wireless rapid diagnostic system for detection of influenza A and B, respiratory syncytial virus (RSV) and group A strep, with new traceability and secure patient health record documentation features and functionality.
The new wireless BD Veritor™ Plus System provides health care providers and laboratorians in physician offices, clinics, hospitals and integrated delivery networks (IDNs) with objective, lab-quality immunoassay test results within minutes. This rapid and accurate solution streamlines the point-of-care (POC) diagnostic workflow and enables providers to quickly review patient results to assist in determining the appropriate treatment in a single consultation. The system can help detect influenza A and B, respiratory syncytial virus (RSV) and group A strep while the patient is still on site, which enables faster decision making for an earlier intervention opportunity.
The BD Veritor Plus System benefits from the demonstrated performance of the legacy BD Veritor System and offers new features to improve quality control (QC) by identifying QC results. The enhanced documentation functionality includes user ID, specimen ID and test kit lot to improve traceability, which decreases manual documentation.
The new BD Veritor Plus System also offers the BD Cloud Connectivity Solution ― a technology that allows users to wirelessly transfer test data securely to an electronic medical record (EMR) or a laboratory information system (LIS) to enable data access across an entire IDN. This can help to reduce potential errors associated with manual documentation and streamlines POC testing workflow to enable physicians to more easily access test results and use the information to determine next steps in patient care.
For more information on the BD Veritor Plus System for use with CLIA-waived flu A+B, RSV and group A strep assays, use request information button below.
Tags:
Date Published: 11th October 2016
E.coli O157 in Raw
ABRx™ Antibiotic Resistance Panel Detects 17







