Lab M ISO Compliant ...
Prolex Staph Xtra - ...
10th November 2007 Content supplied by:
ESwab - New Liquid Based Transport System for Microbiology with Flocked Swabs
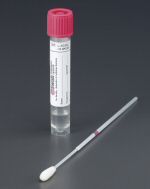
Copan Diagnostics Inc. have launched the ESwab (Elution Swab), a new liquid based transport system for bacteriology swab samples.
This totally original concept for swab transport is made possible through the combination of Copan's patented nylon flock swab invention and a new Liquid Amies format preservation medium. ESwab for the first time provides an open platform for the laboratory to run multiple test analysis. Instead of being limited to one patient's swab, one test, ESwab provides up to TEN identical 100µl aliquots of liquid sample for analysis.
 Unlike traditional fiber wound swabs Copan's nylon flocked swabs provide superior sample absorption and release characteristics. The entire patient's sample is instantly eluted on contact with the ESwab transport medium; there is no need for the operator to rim, ring or vortex the swab.
Unlike traditional fiber wound swabs Copan's nylon flocked swabs provide superior sample absorption and release characteristics. The entire patient's sample is instantly eluted on contact with the ESwab transport medium; there is no need for the operator to rim, ring or vortex the swab.
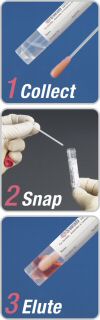
ESwab is extremely easy to use, simply collect the patient's sample with the flocked swab, place the applicator in the ESwab tube, and snap the applicator at the mark, then the sample automatically washes from the swab into the medium: just Collect, Snap and Elute.
Moving the entire patient's swab sample efficiently into liquid phase was not achievable before with traditional fiber winded swabs. This feature provides an important advantage that specimens can be simply and automatically handled using a pipet or pipeting system or transferred to culture plates using the swab, captured by the tube cap, as a convenient inoculation wand.
One millilitre of patient sample in every ESwab tube expands the possible number of first line tests with extra volume in reserve for reflex analysis or sample retention for further investigations. Copan ESwab is a unique multipurpose swab for collection and transport of clinical specimens containing aerobes, anaerobes and fastidious bacteria from the collection site to the testing laboratory.
ESwab is validated in full compliance with CLSI M40-A performance standard; Quality Control of Microbiological Transport Systems. ESwab is demonstrated to maintain the viability of aerobes and anaerobes for up to 48 hours at both refrigerator (4-8ºC) and room temperature (20-25°C) and Neisseria gonorrhoeae for up to 24 hours.
For more information on the ESwab visit www.copanusa.com/products/eswab/
Tags:
Date Published: 10th November 2007
Lab M ISO Compliant Baird-Parker
Prolex Staph Xtra - Rapid







