GMO Food Testing Techniques
GMO in the World
Genetically modified organisms (GMO) were introduced into the food chain over 20 years ago. Today, genetically engineered food production is mainly applied to plants and microorganisms involved in respective processes. Since the use of genetically modified microorganisms is somewhat controversial, the following text refers to genetically modified (GM)-plants only.
The cultivation area for GM crops is continuously growing and development of new GM plants is ongoing. More than 80 % of the cultivated soybeans in the world is genetically modified, cotton is nearly 70% modified, followed by corn and canola at approximately 25 % modified. Although the cultivation of GM crops is concentrated in certain regions (e.g. U.S., Argentina, Brazil), they’re spread globally by trading, transportation and storage either intentionally or inadvertently by contamination of genetically modified organism (GMO) free goods.
The cultivation, use and labelling of GMO is legally regulated in most countries. However, these regulations are not universally consistent. For instance, while the U.S. has an optional labelling of GMO in food products, the European Union (EU) has very strict regulations for the authorization, cultivation and use of GM crops, including an obligatory labelling system.
What is a GMO
To set up appropriate analytical methods, it is important to know what discriminates native and GM variants of a species.
The majority of relevant GM crops on the market are transgenic organisms since they carry artificially introduced genetic material, which isn’t found in their natural gene pool. To insert and activate respective genetic material, a construct of the gene(s) of interest is combined with a promoter and terminator (see sample below). Sometimes marker sequences are also designed and transformed into plant cells. Once introduced, they stabilize and integrate functionally in the plant genome. The locus or specific location of the integration in the genome is relatively random. Functional clones have to be selected and characterized before they are introduced to the market as seed products for agriculture.
The analytical approach to GMO testing
If the consumers´ acceptance of GMO is low (e.g. in Europe), regulators pay less attention to different species of GM crops – or so-called “events”. Thus, the basic analytical approach will check for the general presence of GMO in a sample (also known as GMO Screening). Subsequently, it might be important to know which particular GMO event is contained in a sample (identification), since this has legal consequences. Even a relative amount of GMO in the sample must be determined (also called quantification) in some cases.
How to detect for GMO
Since the difference between the native plant and GM counterpart is based on DNA, the direct method for detection is also DNA based. The best technology to perform DNA analysis in routine labs is real-time polymerase chain reaction (PCR)—a high specificity and sensitivity is combined with affordable, user-friendly handling and easy data interpretation.
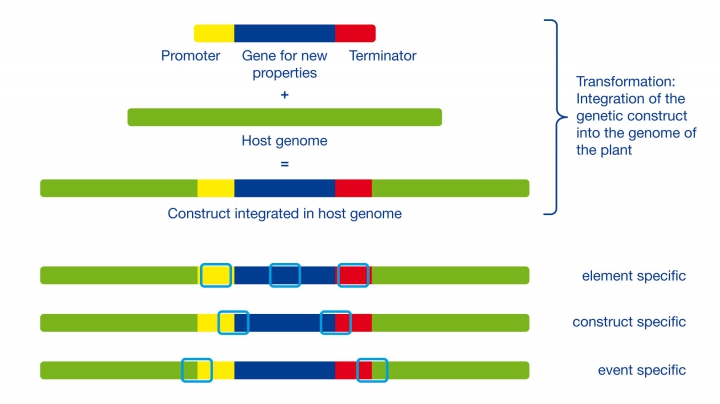
The combination of plant or host genome and integrated construct enables different levels of specificity that can be used for detection in three of the following ways:
Element specific: Single components of the construct like frequently used promoter or terminator sequences can be used. They can appear in several constructs, but also in their natural hosts (e.g. plant viruses).
Construct specific: The transitions between genetic elements of the construct, e.g. the combination of a defined promoter and a gene sequence, are suitable for the detection of distinct constructs. The constructs, however, can be used in different GMO events.
Event specific: The transition of the plant genome to the construct at the integration locus are unique for a GMO event and can be used for identification purposes.
GMO screening, identification and quantification
The purpose of a GMO screening is to detect the maximum of different events with a minimum of analytical effort. Element specific sequences are suitable screening targets since they appear in a large number of GMOs. Respective PCR systems are described in International Organization for Standardization (ISO) norms (ISO 21569, ISO 21570).
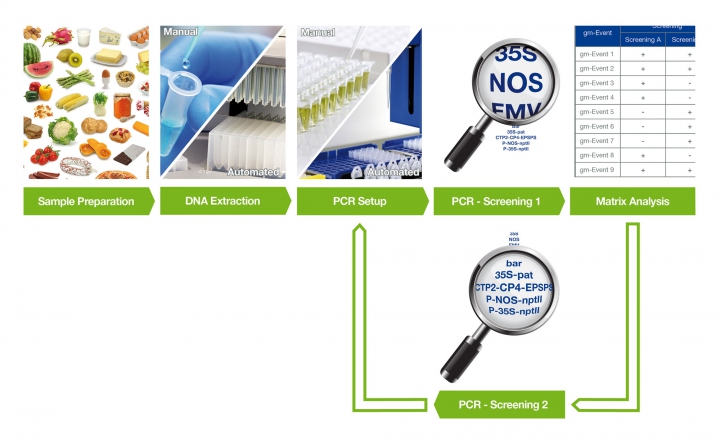
By combining a suitable number of respective targets, a relevant number of events can be identified and helpful information for subsequent identification can be gained. This approach (e.g. described in DIN CEN/TS 16707:2014-12) is based on a screening matrix — a table showing the presence of selected elements with respective GMO events. For instance, plant DNA is extracted from the sample and tested with real-time PCR for the presence of element specific markers selected from the matrix (see chart). The test result allows the exclusion of a majority of possible GMO events in the sample and shows what further screening or identification steps are useful. In addition, the matrix based approach enables further elimination of relevant events, e.g. only canola events.
Screening can be finalized by proving the presence of a distinct GMO event using an event specific target, also known as identification. If the amount of GMO is relevant, these sequences can also be used in combination with taxon (plant) specific targets to perform a quantification— copy numbers of the event and taxon specific sequences are determined using standards with known quantities. The quotient of both numbers is the relative proportion of the GMO in the sample.
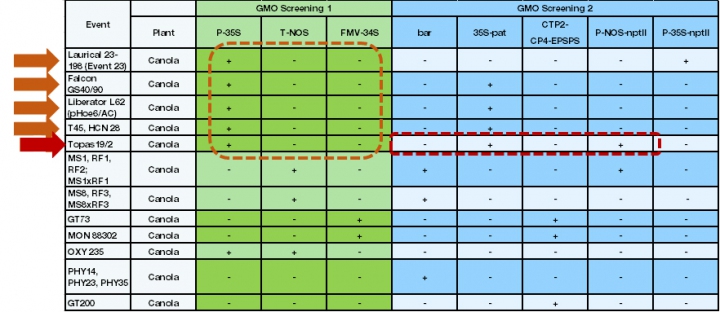
In the example (see chart), since the sample contains canola, only this part of a screening matrix is used for the interpretation of screening results. A basic screening for P-35S, T-NOS and FMV-34S GMOs reveals only the presence of the P-35S element. After the optional exclusion of naturally occurring CaMV (Cauliflower mosaic virus – the natural source for this promoter), further analysis can focus on five distinct events. A second screening analysis with further marker elements reveals the presence of 35S-pat and P-NOS-nptII and the absence of bar, CTP2-CP4-EPSPS and P-35SnptII. Based on the results, the presence of Topas 19/2 is very probable and can be proofed by an event-specific PCR system. Theoretically, a mixture of Topas 19/2 and/or other (see table) events containing only P-35S and 35S-pat is possible.
GMO laboratory analysis
In the laboratory, GMO analysis can easily be established based on available kit systems. DNA extraction is simplified in contrast to work and time intensive traditional methods. Spinfilter or magnetic bead based kits reach at least the efficiency of conventional methods and also allow for automated results. Modern multiplex PCR approaches for GMO screening are optimized concerning handling and interpretation. Kits for l extraction and PCR are validated to ensure maximum reliability of the results.
Since the equipment required is identical, GMO analysis can be integrated in any laboratory already using real-time PCR. Depending on the sample types, proper homogenization tools are required. For laboratories starting GMO analysis, special authorization is not required and a quick return of investment is ensured, since GMO analysis has a high value.
Conclusion
Already, GMO testing is the fastest growing segment In the world. Although in some regions such as Europe the acceptance of GM crops in the food chain is very low, the number of GMO produced continues to grow. In most countries, GMOs are a legally regulated aspect in agriculture, food and food production.
Producers and retailers need reliable information about their raw materials, condiments and products as a basis for correct labelling and to comply with regulations.
Providers of real-time PCR kits have made the previously complex and time-consuming GMO analytical process an easy-to-use real-time application for any food laboratory.
This guide was prepared by the GMO product management of BIOTECON Diagnostics
Get the latest updates in Rapid Microbiological Test Methods sent to your email? Subscribe to the free rapidmicrobiology eNewsletter






