Latest Microbiology News
Microbiome Insights Launches Long-Read Sequencing Services for 16S rRNA Gene and Metagenomics
24 Apr 2024 | Microbiome
Tianlong Human Papilloma Virus (HPV) PCR Kit Detects 18 High Risk HPV Types
23 Apr 2024 | Clinical
Think Outside the Vial with Swab Molecular Quality Controls
23 Apr 2024 | Clinical
RiboFlow® Listeria Twin - Rapid and Simple Molecular Detection
23 Apr 2024 | Food & Beverage
MWE Liquid Media – The Best Solutions for Molecular Diagnostics
23 Apr 2024 | Clinical
Molecular Diagnostics - a rapidmicrobiology Special Focus
23 Apr 2024 | Food & BeverageClinical
New Sample Preparation Solutions to Simplify and Automate Respiratory Diagnostic Testing
22 Apr 2024 | Clinical
HiMedia's Molecular Solutions for Detection of Antimicrobial Resistance
22 Apr 2024 | Clinical
Enhance the Efficiency and Productivity of Your PCR Food Safety Testing
22 Apr 2024 | Food & Beverage
Beckman Coulter’s New CE-Marked Hepatitis Assays at ESCMID Global
17 Apr 2024 | Clinical
High-Quality Pre-Poured Culture Media for Pharmaceutical Applications
16 Apr 2024 | PharmaceuticalCosmetic/Personal Care
Powerful Solution for the Enumeration of Butyric Acid Producing Clostridia (BAPC)
16 Apr 2024 | Food & Beverage
Superior Quality Culture Media for Reliable Media Fill Testing
16 Apr 2024 | Pharmaceutical
HiPetriSlim™: Your Ultimate Solution for Microorganism Testing in Food Samples
16 Apr 2024 | Food & Beverage
Advantages of Compliance with EN17141:2020 in Environmental Monitoring
16 Apr 2024 | Pharmaceutical
BioPerfectus Launches New Diagnostic Solution for SMA (Spinal Muscular Atrophy)
15 Apr 2024 | Clinical
Measurlabs’ Solutions for PFAS Analysis by EU and US Regulations
10 Apr 2024 | Water
Hygiena's Products - A Testament to Excellence in the Dairy Industry
09 Apr 2024 | Food & Beverage
GENE-UP® NUTRAPLEX™ PRO - Molecular Diagnostics Innovation for Nutraceuticals
09 Apr 2024 | Food & Beverage
Visit Puritan® at ECCMID, Booth #E-61 — See You There
09 Apr 2024 | Food & BeverageClinicalLaboratory Equipment
RiboFlow Cronobacter® - Rapid Confirmation/Screening
09 Apr 2024 | Food & Beverage
Win a Limited Edition EVOLVE Manual Pipette Starter Pack
08 Apr 2024 | Laboratory Equipment
TSA Media – What’s Your Pick, What’s the Difference?
08 Apr 2024 | ClinicalPharmaceutical
Revolutionize Your Sampling with Snap-Stick™ Ready-to-Use Culture Swabs
08 Apr 2024 | Laboratory Equipment
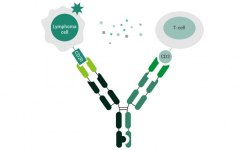
Therapeutic Potential of CD20 x CD3 Bispecific Antibodies
04 Apr 2024 | ClinicalAssay Development
Romer Labs' AgraStrip® Pro Total Aflatoxin WATEX® Test Kit Earns AOAC PTMSM Certification
04 Apr 2024 | Food & Beverage
BIPEA - Proficiency Testing Experts Since 1990s
04 Apr 2024 | Food & BeverageWaterClinicalPharmaceutical
Ziath Mohawk Tube Picker - Fast Large Sample Picking Device
03 Apr 2024 | ClinicalLaboratory EquipmentAssay Development
Eight Surprising Uses of the Growth Direct® System
02 Apr 2024 | Food & BeverageLaboratory EquipmentCosmetic/Personal CareMicrobiome
Understanding Isokinetic Sampling - Why it Matters
02 Apr 2024 | Pharmaceutical