Eight Different Test...
First-Ever Lactic Ac...
14th April 2017 Content supplied by: Mobidiag Oy (HQ)
Mobidiag, Molecular In Vitro Solutions for Syndromic Diagnostics
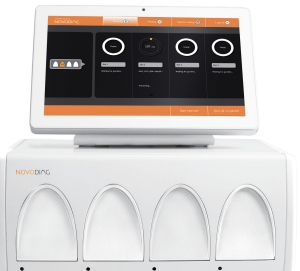 Established in 2000, Mobidiag develops innovative solutions to advance the diagnosis of infectious diseases and serves the European clinical diagnostics market since 2008. Mobidiag addresses both high to medium volume screening with the Amplidiag® Easy platform, bringing the Amplidiag® IVD PCR kits further by automating the workflow, and the upcoming Novodiag® platform (CE-IVD mark pending) showcased at the ECCMID 2017 and associated panels for a fully automated solution and suitable for smaller volumes and labs.
Established in 2000, Mobidiag develops innovative solutions to advance the diagnosis of infectious diseases and serves the European clinical diagnostics market since 2008. Mobidiag addresses both high to medium volume screening with the Amplidiag® Easy platform, bringing the Amplidiag® IVD PCR kits further by automating the workflow, and the upcoming Novodiag® platform (CE-IVD mark pending) showcased at the ECCMID 2017 and associated panels for a fully automated solution and suitable for smaller volumes and labs.
Now available! Amplidiag®, a family of multiplex diagnostic tests and platform for high-volume screening: Amplidiag® H. pylori+ClariR Amplidiag® Stool Parasites Amplidiag® CarbaR+VRE Amplidiag® C. difficile+027 Amplidiag® Bacterial GE
All are compatible with Amplidiag® Easy, automated solution for extraction from primary tubes to PCR plate set-up.
Discover how Mobidiag can support you with rapid and reliable pathogen detection solutions with its upcoming Novodiag®, a turnkey solution for rapid and sensitive on-demand testing. Meet us at booth #100 at ECCMID 2017 (Vienna, Austria)!
Tags:
Date Published: 14th April 2017
Source article link: View
Related news
Eight Different Testing Methods, Only
First-Ever Lactic Acid Bacteria Test







