Sigma GBS™ for Fas...
Case Study Shows How...
4th April 2016 Content supplied by: Charm Sciences Inc.
Charm Peel Plate® Methods Certified for Milk Testing Laboratories
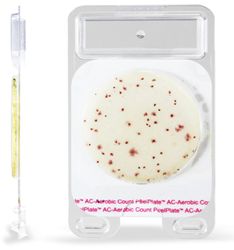 Charm Sciences, Inc. have announced the release of FDA 2400 forms for Charm Peel Plate® methods: Peel Plate AC (aerobic count) and Peel Plate EC (total coliform) tests. The release of these procedures allows industry and state public health milk laboratories to become certified for these indicator tests which can now be used in their routine regulatory testing.
Charm Sciences, Inc. have announced the release of FDA 2400 forms for Charm Peel Plate® methods: Peel Plate AC (aerobic count) and Peel Plate EC (total coliform) tests. The release of these procedures allows industry and state public health milk laboratories to become certified for these indicator tests which can now be used in their routine regulatory testing.
“The release of the 2400 form is a culmination of a lot of work that went into method validation, NCIMS (National Conference of Interstate Milk Shipments) approval, and acceptance into the Pasteurized Milk Ordinance,” stated Bob Salter, VP of Regulatory and Industry Affairs for Charm Sciences. “We have had very positive feedback on these tests, and the dairy industry is welcoming another choice in the simplified methods they can use for bacterial testing.”
The Journal of the AOAC published the method validations that led to the approval of the Charm Peel Plate tests in February of this year. They demonstrated through independent laboratories, that these tests were equivalent to other reference methods. Those articles can be found on the Journal of AOAC International website:
- Peel Plate AC test for detection of Total Aerobic Bacteria in Dairy and Nondairy Products
- Peel Plate EC test for determination of E. coli and Coliform or Total Coliform in Dairy Products
Other Peel Plate tests are available. Visit the Charm website www.charm.com for more information.
Tags:
Date Published: 4th April 2016
Source article link: View
Related news
Sigma GBS™ for Faster Screening
Case Study Shows How to






