Microbiology News: Apr 2017
[VIDEO] How Do You Ensure Your Product Is Truly Allergen Free?
27 Apr 2017 | Food & BeverageOther
Lonza Unveils Agenda for 3rd Annual Global Endotoxin Testing Summit
25 Apr 2017 | Pharmaceutical
Study on IDEXX Legiolert™/Quanti-Tray®, MPN for L.pneumophila
25 Apr 2017 | WaterOther
Solvent-free Detection of Ochratoxin A in Green Coffee and Wheat
25 Apr 2017 | Food & Beverage
How to Choose a Media Preparator/Filler for Your Microbiology Lab
25 Apr 2017 | Food & BeverageClinicalOtherPharmaceuticalVeterinaryCosmetic/Personal Care
Recent Study Evaluates FecalSwab™ for Use in Molecular Tests
24 Apr 2017 | ClinicalOther
Charles River Partners with Microbiologics® to Offer Custom QC Isolates
24 Apr 2017 | OtherPharmaceutical
3rd Annual Charles River QC Micro Workshop: 18 – 21, Sept 2017
24 Apr 2017 | Pharmaceutical
New Listeria Monoclonal Antibodies for Researchers and Kit Manufacturers
21 Apr 2017 | Other
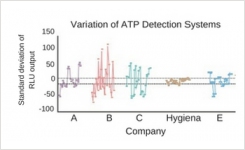
One Reason Why Not All ATP Hygiene Monitoring Systems are the Same
21 Apr 2017 | Food & BeverageOther
Ultra-Rapid Environmental Screening for Legalized Cannabis Growers
20 Apr 2017 | Other
SpeeDx – Molecular Diagnostics Solutions that Go Beyond Detection
18 Apr 2017 | Clinical
UK Acceptance for 24 Hr Detection of P.aeruginosa in Drinking Water
18 Apr 2017 | Water
APAS Independence – The Clever Way to Automate Clinical Microbiology
18 Apr 2017 | Clinical
Sputum Processing for 21st Century - MWE Launches Sigma SP™
17 Apr 2017 | Clinical
First-Ever Lactic Acid Bacteria Test To Gain Independent Validation
14 Apr 2017 | Food & BeverageOther
Mobidiag, Molecular In Vitro Solutions for Syndromic Diagnostics
14 Apr 2017 | ClinicalOther
Eight Different Testing Methods, Only ONE Swab!
14 Apr 2017 | Other
Colibri™ Brings PRECISION and ACCURACY to Microbiology!
14 Apr 2017 | Clinical
Virginia Mold Testing Lab Celebrates 13 Years Serving DC, Maryland and Virginia
13 Apr 2017 | WaterClinicalOther
Quickest, Easiest Test for Raw Pork in Meat and Environmental Samples
13 Apr 2017 | Food & BeverageOther
Scaleable and Versatile Range of Laboratory Water Purifiers
13 Apr 2017 | WaterPharmaceutical
Antimicrobial Susceptibility and Resistance Testing Devices by Liofilchem
11 Apr 2017 | Clinical
FDA Clearance for BacT/ALERT® VIRTUO™ Fully Automated Blood Culture System
11 Apr 2017 | Clinical
Easy EUCAST AST Testing Compliance with ChromaZona
11 Apr 2017 | Clinical
Automated Colony ID on Oxoid Chromogenic Media Using Validated Software
11 Apr 2017 | Food & BeverageClinical
Hygiena's AllerFlow Quickly Detects Gluten in Final Rinse Water Samples
11 Apr 2017 | Food & BeverageOther
Discover VirClia®, Chemiluminescence in Monotest That Adapts to Your Lab
11 Apr 2017 | Clinical
Fast, Accurate QC Microorganisms from Microbiologics
10 Apr 2017 | Clinical
BioFront Technologies Announces US Proficiency Testing Partnership with Fera
10 Apr 2017 | Food & Beverage
bioMérieux Your Expert in Infectious Diseases at ECCMID 2017
10 Apr 2017 | Clinical
AB ANALITICA to Distribute Amplidiag® Diagnostic Tests in Italy
10 Apr 2017 | Clinical
MAST's Extensive Portfolio of Antimicrobial Resistance Products
10 Apr 2017 | Clinical
Special Focus - What's on Show at ECCMID 2017
10 Apr 2017 | ClinicalOther
How To Do Serial Dilutions Better and Now It's Cheaper Too!
06 Apr 2017 | Food & BeverageOther
Ceftazadime/avibactam AST Discs Added to MAST Range
04 Apr 2017 | Clinical
Bacterial Identification Database Updated with Plant Pathogens
04 Apr 2017 | Food & BeverageOther
Drones Fly Specimens Between Swiss Hospital Labs
04 Apr 2017 | Clinical